In patients with high cardiovascular risk, statins are the first-line of therapy. Statins, such as Crestor® and Lipitor®, are economical and effective. However, for those patients who do not have a good response to statins, or those patients with genetic disorders that require aggressive cholesterol lowering, treatment with PCSK9is is an option.
PCSK9is are a new class of cholesterol-lowering drugs that are intended as add-on therapy after maximally-tolerated statins. The initial target patient population includes patients with atherosclerotic cardiovascular disease (ASCVD) and/or heterozygous familial hypercholesterolemia (heFH), a genetic condition that results in high cholesterol levels. There are two PCSK9is on the US market: evolocumab and alirocumab.
A poster presented at the American College of Cardiology (ACC)1 estimated the health benefits of additional LDL-C lowering with alirocumab within the target US patient population. The analysis used patient-level data from the MarketScan Research Database to approximate how many high-risk patients were currently treated with cholesterol-lowering drugs and had low-density lipoprotein cholesterol (LDL-C) levels above the recommended levels. These high-risk patients were identified and scaled to the US population for this analysis, as described in a poster presented at the National Lipid Association (NLA) in 20152. Based on the baseline risk for the patient populations, the analysis estimated the reduction in risk due to an LDL-C reduction with treatment with alirocumab 75 mg and uptitrated to 150 mg if the patient did not achieve the goal LDL-C.
According to the scaling analysis, approximately 8.4 million Americans with ASCVD/heFH are currently on lipid-lowering therapy (LLT) and still have high LDL-C levels. Adding alirocumab to these patients’ current treatment therapies may result in avoiding over 140,000 CV events in one year. Additionally, the number needed to treat (NNT) was 59, representing the number of high-risk patients who would need to be treated with alirocumab to avoid one CV event.
Moreover, for sub-populations of patients with risk above the average ASCVD/heFH patient, NNTs can range from 23 to 133. Full results are shown in the table below.
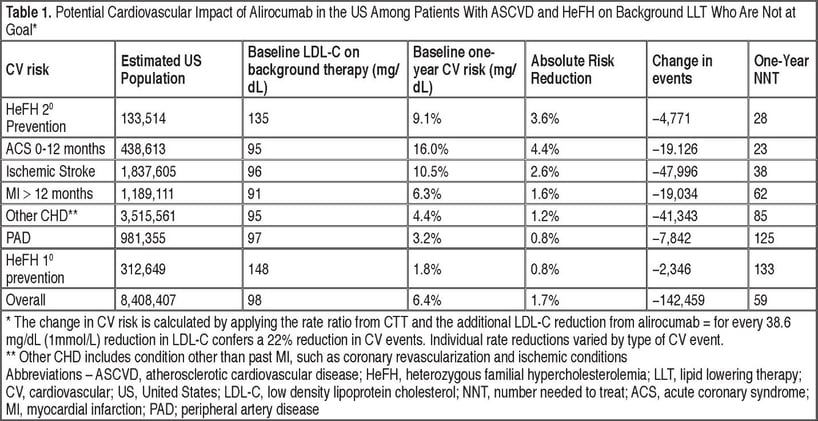
Thus, prioritizing PCSK9i treatment to only those with the highest risk for a cardiovascular event will yield the greatest benefit and avoid the most cardiovascular events. Moreover, the estimated NNTs for many sub-populations are comfortably within acceptable thresholds. This analysis demonstrates the estimated health benefits of alirocumab as add-on to maximally-tolerated statins and provides valuable insight while outcomes-based clinical trial results for alirocumab are still pending.
References:
1. Sanchez R, Nasir K, Klimchak AC, et al. Modeling the US Population Health Benefits of Further Low-Density Lipoprotein Cholesterol Reduction with Alirocumab among Atherosclerotic Cardiovascular Disease or Heterozygous Familial Hypercholesterolemia Patients with Elevated Low-Density Lipoprotein Cholesterol. Poster presented at: The American College of Cardiology 66th Annual Scientific Sessions 2017; March 17-19, 2017; Washington, DC, USA.
2. Gorcyca KM, Khan I, Wadhera R, et al. Prevalence of Atherosclerotic Cardiovascular Disease and Diabetes in the United States. Poster Presented at: National Lipid Association (NLA) Scientific Sessions 2015; June 11-14, 2015; Chicago, IL, USA.



