All Insights Article Modeling the "Difficult to Model" Disease: Considerations for Evaluating the Cost-Effectiveness in Rare Diseases
Modeling the "Difficult to Model" Disease: Considerations for Evaluating the Cost-Effectiveness in Rare Diseases
Modeling the "Difficult to Model" Disease: Considerations for Evaluating the Cost-Effectiveness in Rare Diseases
Axtria’s real-world evidence/health economics and outcomes research (RWE/HEOR) team combines its extensive rare disease domain knowledge with the latest AI/ML techniques.

Defining Rare Diseases and Their Epidemiology
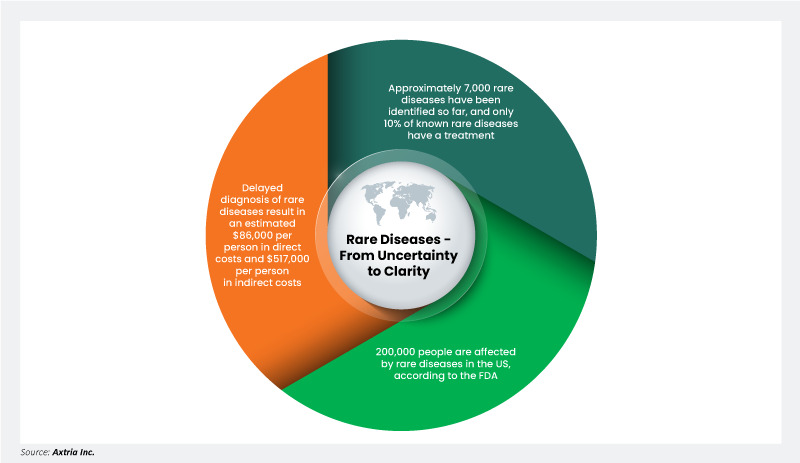
Rare diseases are broadly defined as those afflicting only a small proportion of the population. However, the definitions used for rare diseases are often inconsistent due to a lack of data about their natural history, drug development bottlenecks, and epidemiological data that varies from country to country. For instance, in Europe, rare diseases affect a small number of people, typically fewer than 1 in 2,000.1 However, in the United States, the Food and Drug Administration (FDA) defines a rare disease as affecting fewer than 200,000 individuals.2
Approximately 7,000 rare diseases have been identified so far, and only 10% of known rare diseases have a treatment.3,4 These diseases place a significant economic and humanistic burden on individuals and the healthcare system. According to a study by the EveryLife Foundation, delayed diagnosis of rare diseases resulted in an estimated $86,000 per person in direct costs and $517,000 per person in indirect costs.5
Challenges and Recommendations for a Cost-Effectiveness Analysis (CEA) of Rare Diseases
Conducting a CEA for rare diseases is exceedingly difficult due to a scarcity of data on epidemiology, burden, disease progression, patient characteristics, and treatment effects. These challenges include inadequate trials, heterogeneity in the trial population, and endpoints that prevent the potential use of meta-analyses to support economic model development. Policymakers and the health technology assessment (HTA) bodies recognize that drugs for rare diseases cannot be evaluated using traditional CEAs. HTA bodies have issued guidance and recommendations to navigate these complexities.
- National Institute for Health and Care Excellence (NICE)
For reimbursement decisions in the United Kingdom, NICE has established a special appraisal program called Highly Specialized Technology (HST)6 to evaluate innovative cell and gene therapies for rare diseases. The HST program specifically focuses on technologies that are used to diagnose and treat a very small population – typically fewer than five per 10,000 people. The HST appraisal program removes criteria regarded as redundant with the development of one-off cell and gene therapies (i.e., the requirement that the disease be "chronic and severely disabling" and that the treatment must be "lifelong" in nature). HST appraisals typically consider only the rarity of the disease, its impact on health, and the lack of available treatments. As of April 2017, NICE has set a maximum additional quality adjusted life years (QALY) approval threshold of £300,000 for HSTs, which is ten times higher than the approval threshold (i.e., £20,000 to £30,000 per QALYs) for non-specialized treatments.7
- Affordability considerations: Although HST appraisals assess the cost-effectiveness of treatments, traditionally they do not assess affordability in terms of overall cost to the National Health Service (NHS) for the commissioning of a treatment. Novel rare disease interventions could be quite expensive and commissioning them may impact NHS budgets. However, since April 2017, NICE has included flexibility for a phased introduction of a novel treatment that may not significantly affect the NHS budget. This incremental strategy, usually restricted to three years, tackles issues pertaining to immediate fiscal pressures.7
- Collaboration for affordability: In 2016, the Public Accounts Committee advised NICE and NHS England to collaborate to incorporate affordability into the decision-making process for specialized treatments. This collaboration aims to strike a balance between granting access to cutting-edge therapies for uncommon medical conditions and efficiently controlling healthcare expenses.8
- Affordability considerations: Although HST appraisals assess the cost-effectiveness of treatments, traditionally they do not assess affordability in terms of overall cost to the National Health Service (NHS) for the commissioning of a treatment. Novel rare disease interventions could be quite expensive and commissioning them may impact NHS budgets. However, since April 2017, NICE has included flexibility for a phased introduction of a novel treatment that may not significantly affect the NHS budget. This incremental strategy, usually restricted to three years, tackles issues pertaining to immediate fiscal pressures.7
- Institute for Clinical and Economic Review (ICER)
In 2017, the independent non-profit organization ICER made significant modifications to value-based frameworks (VAFs) based on meaningful insights gained from discussions with patient groups and other stakeholders. The aim was to help the US strike the right balance between encouraging innovations, ensuring access, and maintaining affordability through a collaborative approach between stakeholders and public engagement.
- Adaptive analysis and flexible thresholds: For rare diseases, ICER seeks to produce a cost-effectiveness (CE) model for every new treatment and highlight additional uncertainty in translating patient outcomes into QALYs or equal value of life year gained (evLYG) measures. Further, ICER adapted a willingness-to-pay (WTP) threshold from $50,000 to $500,000 per QALY without weight to QALY gains or baseline disease severity.9
- Consistent evidence rating: In assessing any serious ultra-rare disease condition, ICER ensures the same Evidence-Based Medicine (EBM) matrix for rating evidence, applying the same standards regardless of the rarity of the disease. ICER acknowledges the challenges in conducting randomized trials, validating surrogate outcome measures, and obtaining long-term safety and clinical benefit data for ultra-rare diseases.9
- Adaptive analysis and flexible thresholds: For rare diseases, ICER seeks to produce a cost-effectiveness (CE) model for every new treatment and highlight additional uncertainty in translating patient outcomes into QALYs or equal value of life year gained (evLYG) measures. Further, ICER adapted a willingness-to-pay (WTP) threshold from $50,000 to $500,000 per QALY without weight to QALY gains or baseline disease severity.9
Value-Based Frameworks for Assessment of the Value of Drugs in Rare Diseases
To ensure that patients can access novel treatments for rare diseases, more robust VAFs are being introduced by the HTAs. For rare diseases, broad value-based approaches that are followed across HTAs include1:
- Multi-Criteria Decision Analysis (MCDA)
MCDA is a decision-making tool that can help address the challenges of CEA in rare diseases.10 It allows the consideration of multiple criteria, including costs, effects, and other factors relevant to rare diseases. An intervention's composite score is produced using a matrix that contains several components, such as unmet needs, disease severity, and cost-effectiveness. First, all pertinent criteria for value are identified by a multidisciplinary team (appraisal criteria). The next step involves prioritizing the criteria by assigning weights based on their relevance to the evaluated treatment. The intervention's performance is then evaluated against each criterion and given a score. The ultimate outcome is an aggregate score, determining the most desirable treatment to fund or creating a ranking list of treatments.1 - QALY weighting
Replaces the existing “end-of-life” QALY modifier with a severity modifier that assigns a higher weight to conditions with the most significant health impact. - Variable Increment Cost Effectiveness Ratio (ICER) threshold
In this approach, ICER thresholds are based on predetermined appraisal criteria or societal preferences (such as prevalence, severity, or social values). An intervention is judged to be cost-effective when its ICER is below the threshold. - No economic evaluation
In this approach, an intervention is evaluated not by cost-effectiveness measures but by considering evaluation criteria such as efficacy, safety, or added therapeutic value compared to that of the standard of care (SoC). - Standard economic evaluation
Under this standard approach, the intervention's cost and effectiveness are compared against that of the SoC. The outcome of the analysis is ICER, one must pay to gain a unit of health benefit over SoC.
Axtria's Approach to Address Challenges in Cost-Effectiveness Analyses for Rare Diseases
HTA bodies across the globe are moving towards a more systematic, value-based framework for rare disease drug assessment. Still, the paucity of data about the clinical impact of treatment, quality of life effect, resource utilization, and more continues to pose unique challenges.
Axtria works in close partnership with pharmaceutical companies and key opinion leaders (KOLs) to develop fully customized, innovative, clinically sound, and scientifically robust solutions to address some of these challenges.
- Limited patient population
Significantly, lower disease prevalence restricts the number of patients available for observational studies or clinical trials, resulting in higher levels of heterogeneity in the sample and, thus, higher uncertainty in the treatment effectiveness. Additionally, the trial population might not perfectly align with the population of interest in each market.
Implementation of creative statistical approaches like Bayesian analysis can help account for small sample sizes. With advanced artificial intelligence/machine learning (AI/ML) techniques leveraging clinical algorithms and data points such as biomarkers, lab tests, and procedures, one can identify a population of interest more accurately. That, in turn, can result in larger sample sizes. Also, if any published evidence about different patient characteristics and their disease progression is available or can be teased from discussions with clinical experts or KOLs, a pseudo-patient population can be generated and further simulated to provide additional data points. - Single arm trials
Due to the nature of rare or ultra-rare diseases and the lack of alternate treatments, some clinical trials are designed without a comparator arm. Real-world data sources like patient registries, claims data, patient-reported outcomes (PROs), chart reviews, and discussions with KOLs can be used to create synthetic comparator arms.
- It is essential to emphasize the significance of patient engagement and utilization of patient registry data in the economic modeling of rare diseases, mainly where clinical data is inadequate, which is consistent with the recommendations of the NICE HST submission.11 Also, surveys about patients’ and caregivers’ productivity, daily activities, and psychological distress can be employed as clinically meaningful evidence for model developments of rare diseases.
- Patients' registry data can provide clinical data for the comparator arm (i.e., standard of care) to overcome the challenges associated with the lack of clinical data for rare diseases. Access to long-term registry data can provide an understanding of the natural history data of a rare disease; that data can be used as a surrogate for standard of care in the absence of placebo data.11
- It is essential to emphasize the significance of patient engagement and utilization of patient registry data in the economic modeling of rare diseases, mainly where clinical data is inadequate, which is consistent with the recommendations of the NICE HST submission.11 Also, surveys about patients’ and caregivers’ productivity, daily activities, and psychological distress can be employed as clinically meaningful evidence for model developments of rare diseases.
- Insufficient data on disease progression
Sometimes rare diseases have early (i.e., childhood) onset of illness. It is essential to involve patients, caregivers, advocacy groups, and clinical professionals early in economic model development to ensure adequate model structure and capture all costs and clinical consequences.
In another scenario, there may be long durations of misdiagnoses for patients with rare diseases. While developing the model structure, published data can be used to define different health states. Although there is a lack of data on disease progression, discussions with KOLs can be held to identify a biomarker or key metric of the disease that can be used to estimate disease severity.
A combination of novel predictive modeling techniques and statistical clustering can segment the patient journey into different stages or health states. Propensity models can be developed on longitudinal data, if available, using techniques like multinomial logistic regression (or other classification algorithms) to calculate the probability of a patient progressing from one state to another. - Lack of high-quality utility estimates
Limitations in the designs of clinical trials, registries, or other observational studies can lead to uncertainties in the health-related quality of life (HRQOL) information and the resulting estimates of QALYs. Axtria consults on identifying appropriate PRO instruments to be used in the clinical trial. Since many of the rare diseases are debilitating, patients require constant care. It becomes important to account for the impact of the disease on caregivers’ quality of life as well as to fully understand the humanistic burden of the disease and the benefit of treatment.
Conclusion
Limited quality data on disease burden, disease progression, patient characteristics, treatment effects, and stringent policies have been key hindrances in drug approvals and reimbursements for rare diseases. Specialized programs like NICE's HST and initiatives to streamline assessment processes and incorporate affordability considerations are critical steps forward. Institutions like ICER are also adapting their approaches by performing flexible thresholds and are helping to navigate the complexities of cost-effectiveness analysis for rare diseases.
HTA bodies increasingly acknowledge the need for more robust value-based frameworks to assess rare disease treatments. These flexible approaches have created opportunities to use advanced predictive modeling techniques in cost-effectiveness analyses.
Axtria’s real-world evidence/health economics and outcomes research (RWE/HEOR) team combines its extensive rare disease domain knowledge with the latest AI/ML techniques. The cost-effectiveness models Axtria builds helps HTA bodies make informed reimbursement decisions. Our unique approach blends real-world evidence expertise with robust strategic and analytical skills, empowering life science companies to excel in generating RWE and strengthening the value proposition of their products. By improving the rare disease models, we constantly endeavor to create better patient outcomes.
References
- Blonda A, Denier Y, Huys I, Simoens S. How to value orphan drugs? A review of European value assessment frameworks. Front Pharmacol. 2021 May 12;12:631527. doi: 10.3389/fphar.2021.631527. PMID: 34054519; PMCID: PMC8150002.
- Rare diseases at FDA. United States Food and Drug Administration. Accessed February 28, 2024. https://www.fda.gov/patients/rare-diseases-fda
- Medical products for rare diseases and conditions. United States Food and Drug Administration. Accessed February 28, 2024. https://www.fda.gov/industry/medical-products-rare-diseases-and-conditions
- Rare Disease Cures Accelerator. United States Food and Drug Administration. Accessed February 28, 2024. https://www.fda.gov/drugs/regulatory-science-research-and-education/rare-disease-cures-accelerator
- EveryLife Foundation. The cost of delayed diagnosis in rare diseases: A health economic study. EveryLife Foundation, The Lewin Group. September 14, 2023. Accessed February 28, 2024. https://everylifefoundation.org/delayed-diagnosis-study/
- Highly specialised technologies guidance. National Institute for Health and Care Excellence (UK). Accessed February 28, 2024. https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-highly-specialised-technologies-guidance
- Powel T. NICE appraisals of rare diseases. House of Commons Library. March 12, 2019. Accessed February 28, 2024. https://researchbriefings.files.parliament.uk/documents/CDP-2019-0022/CDP-2019-0022.pdf
- Bate A. NHS commissioning of specialised services. June 9, 2017. Accessed February 28, 2024. https://researchbriefings.files.parliament.uk/documents/CBP-7970/CBP-7970.pdf
- Modifications to the ICER value assessment framework for treatments for ultra-rare diseases. Institute for Clinical and Economic Review. November 2017. Accessed February 28, 2024. https://everylifefoundation.org/delayed-diaghttps://icer.org/wp-content/uploads/2020/10/ICER-Adaptations-of-Value-Framework-for-Rare-Diseases.pdf nosis-study/
- Postma MJ, Noone D, Rozenbaum MH, et al. Assessing the value of orphan drugs using conventional cost-effectiveness analysis: Is it fit for purpose? Orphanet J Rare Dis. 2022 Apr 5;17(1):157. doi: 10.1186/s13023-022-02283-z. PMID: 35382853; PMCID: PMC8981887.
- Pearson I, Rothwell B, Olaye A, Knight C. Economic modeling considerations for rare diseases. Value Health. 2018 May;21(5):515-524. doi: 10.1016/j.jval.2018.02.008. Epub 2018 Apr 17. PMID: 29753347.

Complete the brief form to download the white paper

Javed Shaikh
Javed is a Director in the RWE/HEOR practice at Axtria with 15 years of experience in RWE/HEOR. He is an experienced subject matter expert in generating and using the evidence (including RWE) to create differentiated, compelling value propositions to payers, providers using Value Evidence, Pricing, and Market Access strategies.

Sandeep Tripathi
Sandeep is Associate Director in the RWE/HEOR practice at Axtria, with over 11 years of experience in developing health-economic models, conducting trial data analyses, and synthesizing evidence. He gathers his experience from working with multiple clients solving varied problems across therapeutic areas such as cardiovascular diseases, diabetic retinopathy, oncology, and rare genetic diseases.

Complete the brief form to download the white paper
Related insights
Stay current on topics you care about
Recommended insights

Article







.jpg?width=690&height=468&name=AI-ML-in-life-sciences%20(1).jpg)