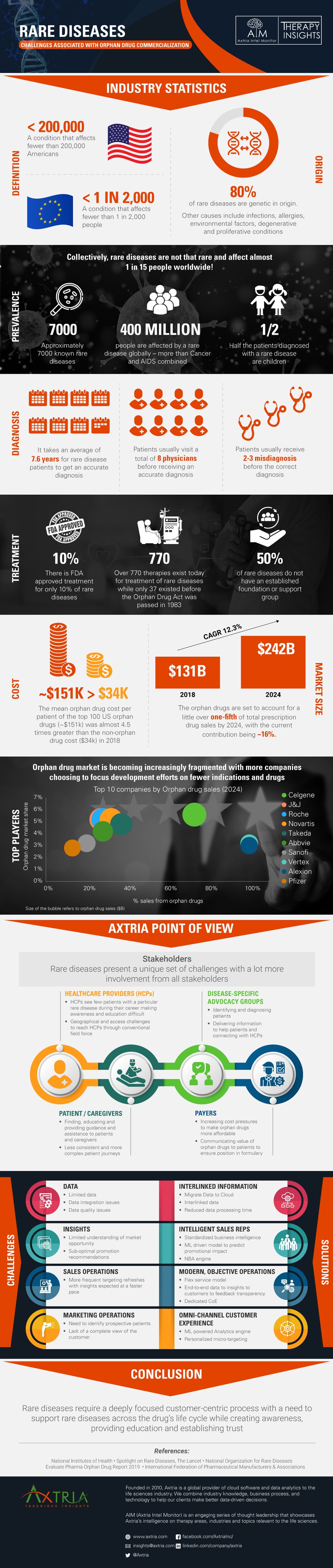
Rare Diseases - Challenges Associated With Orphan Drug Commercialization
There are about 7,000 known rare diseases that collectively affect around 1 in 15 people worldwide, about half of them children. Given the limited understanding of many of the rare diseases, patients struggle with getting the right diagnosis and the right treatment, with an FDA approved treatment available for only 10% of rare diseases.
The commercialization process of orphan drugs differs from that of traditional drugs with a lot more involvement from multiple stakeholders, each with their own set of unique challenges. To succeed in this environment, pharma companies need to focus on creating awareness and building trust among patients while fostering a culture of patient-centricity.
Check out the infographic below to learn about Rare Diseases: